|
You would be thinking why…!!! Well, just because the outermost shell of noble elements is filled, they are stable. Atoms are Unstable and Highly ReactiveĮxcept for noble elements, all other atoms are unstable and highly reactive. In addition, atoms are spherical in shape. On the other hand, electrons are held together to the atomic nucleus by electromagnetic force. They are primarily made up of a nucleus (proton and neutron) and electrons, where the nucleus of an atom is held together by nuclear force. Just to let you know that a picometer is equal to one trillionth of a meter. As per the available scientific data, atoms are approximately 100 picometers across. That’s why they are neither visible to the naked eye nor to magnifying microscopes. They are that much small that their behavior cannot be predicted using classical physics. 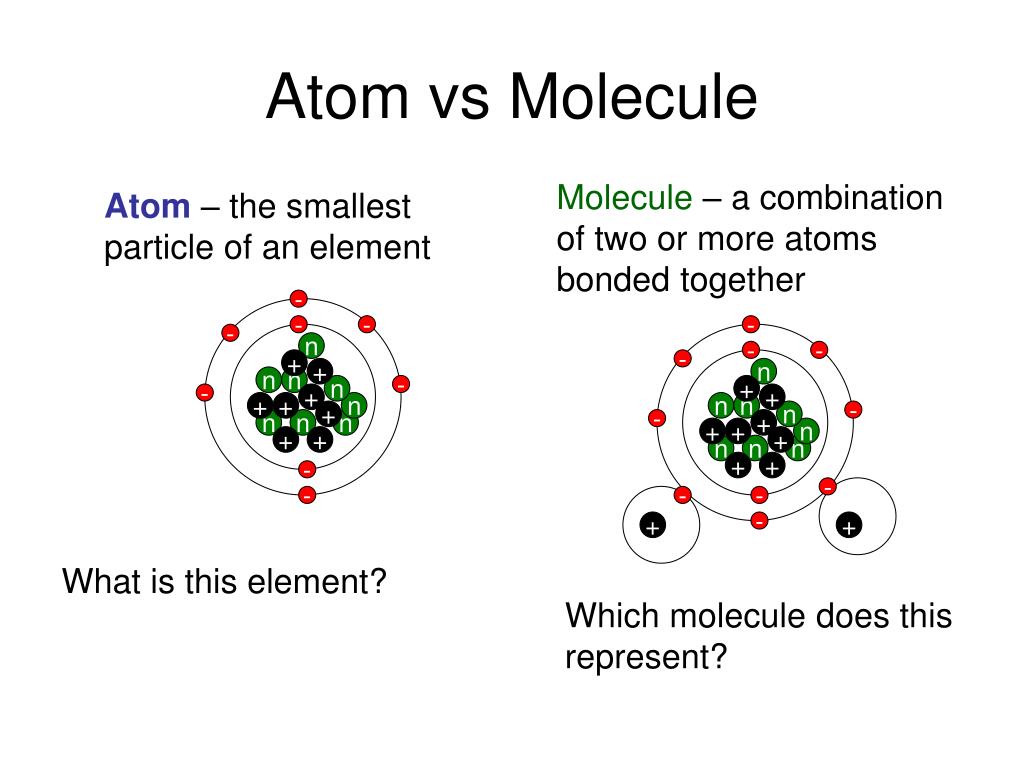
Some examples of molecules are water (H 2O) and Carbon dioxide (CO 2)īy definition, an atom is the smallest constituent unit of any element. Some examples of atoms are Hydrogen (H) and Oxygen (O). 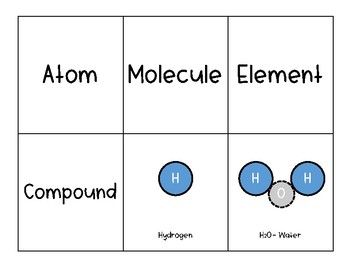
Molecules are visible through a magnifying microscope but not through the naked eye. 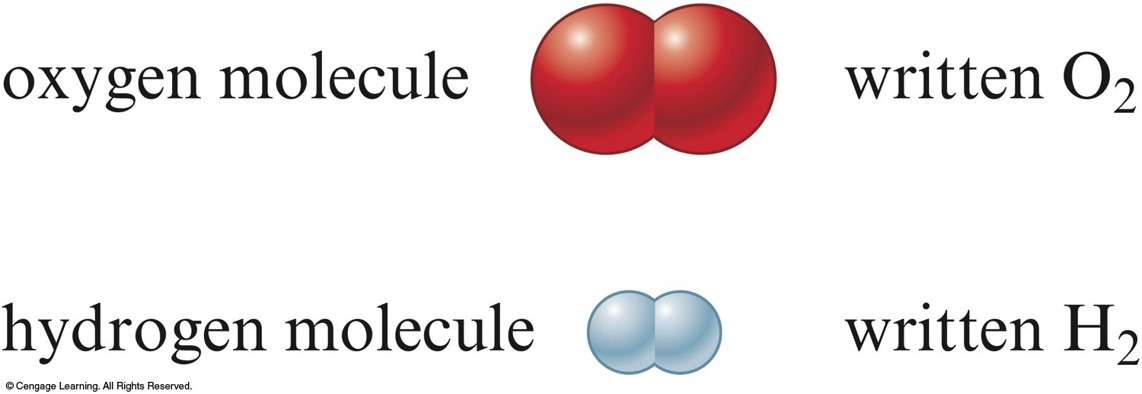
It is made up of two or more similar as well as different elements.Įxcept for noble elements, all other atoms are highly reactive in nature.Ītoms are neither visible to the naked eye nor to magnifying microscopes. It is made up of electrons, protons, and neutrons. Molecules are formed to attain stability and hence can stay freely. An atom is the smallest constituent unit of any matter or element.Ī molecule is a combination of two or more atoms.Ītoms are mostly unstable and hence cannot stay freely.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
